FDA Seeks Feedback on Opioid Blister Packs
/By Donna Gregory Burch
I’ve been hoarding a bottle of oxycodone tablets from my back surgery last year. I probably shouldn’t admit it, but I keep them tucked away in the back of a bathroom drawer in case my chronic back and fibromyalgia pain gets so intense that I need something stronger than the tramadol or medical marijuana I normally use for daily pain.
I’m doing exactly what the Food and Drug Administration doesn’t want me to do. In fact, if an FDA proposal is adopted, there will be far fewer patients like myself with stashes of leftover opioids.
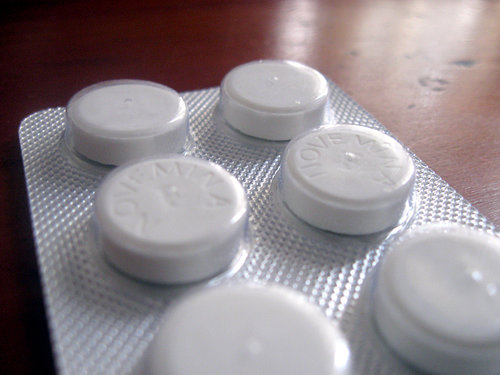
The FDA is soliciting public comments on a proposal to require drug makers to manufacture certain opioids in “blister packs” for acute pain situations such as patients recovering from injuries and minimally-invasive surgery. Opioids listed in the FDA’s Federal Register notice include hydrocodone, tramadol, oxycodone and codeine.
In a nutshell, the FDA would like doctors to have the option to prescribe 5, 10 or 15-count opioid tablets in foil wrappers, similar to those annoyingly difficult-to-open packages used for cold-and-flu medications.
If you’ve ever been sick as a dog with a cold and used all of your strength to wrestle a Dayquil capsule out of its foil wrapper, then you know the type of packaging I’m talking about.
The FDA’s proposal is intended to reduce the number of unused opioids that could wind up in the hands of children and others who might abuse or divert them. Based on published studies, most “opioid naïve” patients who are new to opioids don’t actually take all of the tablets prescribed to them for acute pain. The FDA believes all of those leftover pills stashed in medicine cabinets could be contributing to the so-called opioid crisis.
“In the post-surgical setting, following several common minimally or less-invasive surgical procedures, most opioid-naïve adults who used an opioid analgesic appeared to use only one to three days’ worth, or 15 or fewer, opioid analgesic tablets or capsules despite receiving prescriptions exceeding the number they used,” reads the FDA’s notice for public comment.
“(We) anticipate that utilization of these fixed-quantity unit-of-use blister package configurations would substantially reduce the quantity of opioid analgesics dispensed per prescription compared to the status quo.”.
Obviously, physicians already have the authority to prescribe smaller quantities of opioids, but the FDA says these blister packages would encourage more rational “right-size” opioid prescribing. The notice stipulates that blister packs would not be required and that other options would still be available.
Other potential benefits of blister packaging cited by the FDA include:
Reducing accidental childhood poisoning
Being able to include additional labels on packaging on how to safely use and dispose of opioids
Making it easier for patients and caregivers to determine if a third party has taken their opioid medication
The FDA is soliciting comments on its proposal through July 30. Read the public comment notice here. The notice includes instructions for submitting comments online, by email or in paper form.
So, what do you think? Would prescribing opioids in blister packaging for acute pain help curb the opioid crisis? Am I the only person who breaks out in a sweat when I have to pry medications from that kind of packaging? Share your thoughts (and struggles) in the comments!
Donna Gregory Burch lives with fibromyalgia and chronic Lyme disease. Donna covers news, treatments, research and practical tips for living with fibromyalgia and Lyme on her blog, FedUpwithFatigue.com. You can also find her on Facebook and Twitter.
Donna is an award-winning journalist whose work has appeared online and in newspapers and magazines throughout Virginia, Delaware and Pennsylvania. She lives in Delaware with her husband and their many fur babies.