Magnetic Implant Could Someday Deliver Medication
/By Pat Anson, Editor
Over the years scientists have developed a variety of drug delivery systems designed to help patients take medications more safely – from pumps to implants to pills made with abuse-deterrent formulas.
Researchers at the University of British Columbia have now developed one of the strangest ones yet -- a magnetic drug implant -- that could offer an alternative for pain patients who don’t like pills or injections and fear the idea of having a pain pump installed.
“This could one day be used for administering painkillers, hormones, chemotherapy drugs and other treatments for a wide range of health conditions. In the next few years we hope to be able to test it for long-term use and for viability in living models,” said Mu Chiao, PhD, a professor of mechanical engineering at UBC.
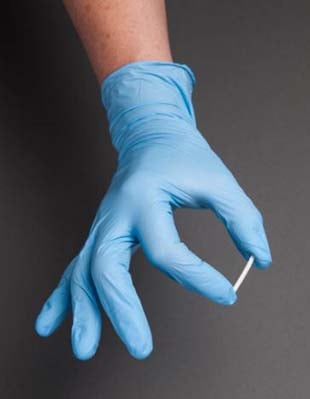
COURTESY UBC
The magnetic device – a silicone sponge with magnetic carbonyl iron particles wrapped in a polymer layer – is just six millimetres (about a quarter of an inch) in diameter. The drug is injected into the device and then surgically implanted in the area being treated.
Researchers tested the device on animal tissue in the lab using the prostate cancer drug docetaxel. They found that it was able to deliver the drug on demand even after repeated use. The drug also produced an effect on cancer cells comparable to that of freshly administered docetaxel, proving that drugs stored in the device remain effective.
Passing a magnet over the patient’s skin activates the device by deforming the sponge and triggering the release of the drug into surrounding tissue.
The University of British Columbia released this short video to show how it works:
“Drug implants can be safe and effective for treating many conditions, and magnetically controlled implants are particularly interesting because you can adjust the dose after implantation by using different magnet strengths. Many other implants lack that feature,” said Ali Shademani, a PhD student in the biomedical engineering program at UBC, who was lead author of a study published in the journal Advanced Functional Materials.
Implants such as Probuphine – which was approved last year by the Food and Drug Administration to treat opioid addiction -- cannot be adjusted to deliver different medication levels once they are implanted.
The UBC researchers say actively controlling drug delivery is important not only for treating pain, but for conditions like diabetes, where the required dose and timing of insulin varies from patient to patient.
“This device lets you release the actual dose that the patient needs when they need it, and it’s sufficiently easy to use that patients could administer their own medication one day without having to go to a hospital,” said co-author John Jackson, a research scientist in UBC’s faculty of pharmaceutical sciences.