The CDC and Profit Driven Drug Testing
/(Editor’s Note: As part of its draft opioid prescribing guidelines, the Centers for Disease Control and Prevention (CDC) recommends that physicians “use urine drug testing before starting opioid therapy and consider urine drug testing at least annually to assess for prescribed medications as well as other controlled prescription drugs and illicit drugs.” Mark Collen is an expert in this field and has submitted the following to the CDC.)
By Mark Collen, Guest Columnist
There are a number of conflating factors which have led to the CDC‘s recommendation for urine drug testing (UDT) in pain management -- none of which have to do with quality patient care. These include the federal government‘s complicity in the overprescribing of drugs, the unethical marketing of OxyContin by Purdue Pharma, the profit motive which drove drug testing patients in pain, and physicians‘ fears and inability to effectively treat pain.
We live in a drug addled society with Americans consuming record amounts of prescription drugs, including opioids. In other words, American healthcare providers are exceptional at writing prescriptions. Someone needs to tell them to, “Just say no.”
The pharmaceutical industry‘s direct-to-consumer advertising (DTCA) has driven demand for drugs and has contributed to the escalating cost of prescription medications. Both Congress and the FDA are largely responsible for this overprescribing public health issue since they have allowed DTCA to exist and spread.
What has not been investigated is the influence the incessant drug ads have had on the American culture and more importantly, the American psyche in terms of one‘s views on drug use, prescription or otherwise. Almasi and colleagues write, “DTCA pushes a ‘Brave New World‘ where if anything unpleasant should somehow happen, why, there‘s always [the sedative] soma to give you a holiday from the facts.” DTCA may influence the abuse of opioids and other drugs.
The pharmaceutical company Purdue Pharma was responsible for one of the most deceptive marketing campaigns in history with their medication OxyContin, a timed-release oxycodone. Art Van Zee writes, “A consistent feature in the promotion and marketing of OxyContin was a systematic effort to minimize the risk of addiction in the use of opioids for the treatment of chronic non–cancer-related pain.” As a result, OxyContin became the poster boy for prescription drug abuse and Purdue was fined $600 million to settle criminal complaints. That has had little impact on the Sackler family, Purdue‘s owners, with Forbes estimating their worth at $14 billion.
Although Purdue is not completely responsible for the current opioid abuse epidemic, they certainly have played a meaningful role. In addition, a congressional investigation suggested that the pharmaceutical industry has promoted opioid sales through a number of organizations including the American Pain Society and the American Academy of Pain Medicine, both of which write treatment guidelines.
As opioid abuse and overdose deaths began to rise, so did the drug testing industry within pain management. Drug testing labs used a similar strategy as the pharmaceutical industry in promoting their wares and money was used as an incentive to drug test.
In my 2012 article entitled, “Profit-Driven Drug Testing,” I presented Medicare data which showed a meteoric rise in drug testing:
“A deeper examination finds that between 2000 and 2009, the total number of CLIA-waived drug tests paid for by Medicare and conducted at physicians‘ offices increased approximately 3,172,910%; with 101 tests conducted in 2000 and 3,204,740 in 2009. Furthermore, during that same time period and within the specialty of anesthesiology, CLIA-waived drug tests increased 63,687,900%.”
The annual cost of drug testing in pain management is estimated at $2 billion per year. Unfortunately, that may be a gross underestimate since no study has ever evaluated the indirect costs of patient harm or harming the therapeutic patient-provider relationship—likely the most important aspect of pain management. A November, 2014 article in the Wall Street Journal reported that some physicians are making more money from drug testing patients than treating them.
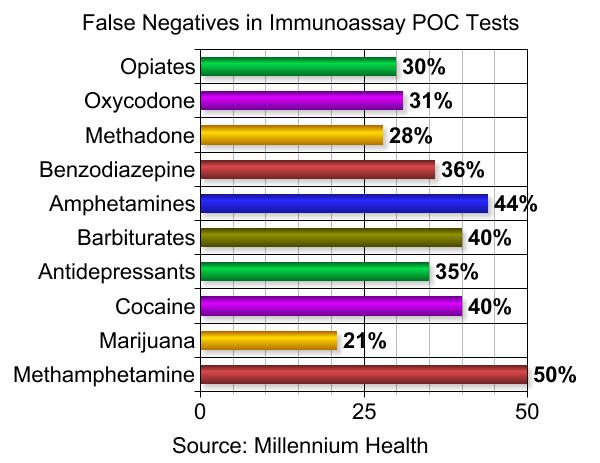
Not unlike big pharma, there appears to be a dearth of integrity in the drug testing industry. Millennium Health, the largest drug tester in pain management, was recently fined $256 million by the U.S. Department of Justice and then filed for bankruptcy. This led to the discovery that the founders took $1.3 billion out of the business in 2014.
Ameritox, the second largest drug tester, actually paid physicians to drug test their patients, and as a result was fined $16.3 million by the Justice Department. Calloway Laboratories is yet another drug testing lab that was prosecuted and it is going out of business.
This author asked Debra Maul, a whistleblower in the laboratory industry, for her comment on UDT in pain management for this paper. Debra wrote, “Personally, I believe it‘s all about the money. When I entered the laboratory business in 2003, it was very difficult to get physicians to test their patients. In 2007, when Millennium entered the industry with the POCT (point of care tests) business model, pain doctors significantly increased their patient testing, I believe, because they could make money on in-office testing. New labs were popping up everywhere promoting this business model.”
She continued, “If you look at the information the WSJ obtained from CMS regarding Medicare reimbursements for UDT, reimbursements for simple UDTs grew significantly from 2007 until reimbursements were cut in 2010. Then in 2011 and 2012, high tech drug testing took a big jump, I believe, due to laboratories promoting in-office analyzers and other high-tech testing equipment to doctors, so they could continue billing for UDTs. It will be interesting to see what happens with in-office testing and the entire UDT market, with the significantly reduced reimbursements this year.”
What follows is a list of drug testing labs and the amount they were reimbursed by Medicare for urine drug testing in 2012. These numbers come from Medicare‘s website and were provided by Debra Maul:
- Millennium: $109,031,768
- Ameritox: $99,553,258
- Aegis: $36,140,368
- Alere: $16,937,116
- AIT: $13,845,880
- Dominion: $12,551,313
- Calloway: $6,918,972
To complicate matters, healthcare providers do a poor job at treating chronic pain and opioids may not be the best choice for the long-term treatment of chronic, non-acute, non-malignant pain. Moreover, insurers have been known to reimburse for pain medication and not physical therapy. It is likely that a number of clinicians prescribe opioids because they don‘t know what else to do, and then perform random drug tests in hopes of mitigating any damage they may cause, but it does not work. There is a great need to develop and test cost effective, alternative interventions to pharmacotherapy for the treatment of chronic pain and illness in the primary care setting.
The overriding factor in this historical perspective is the consistent and negative impact industry has on medicine and in this case, pain medicine. A good example of how close the drug testing industry gets to individuals who write guidelines, please go to this link and note the presenter is the lead author of the CDC guideline (Dr. Deborah Dowell) and a conference sponsor is a drug testing lab (Ameritox).
There is no question that industry has a negative influence on medicine. Stamatakis and colleagues write, “The industry has created means to intervene in all steps of the processes that influence healthcare research, strategy, expenditure and practice. These include evidence base production, evidence synthesis, understanding of harms issues, cost-effectiveness evaluation, clinical guidelines formation, healthcare professional education and direct influences on healthcare professional decisions.”
Urine drug testing in pain management costs an estimated $2 billion per year and there is no proof of efficacy. Moreover, it may cause patient harm and harm to the patient-provider relationship and thus increase healthcare costs even further.
The desire for profits likely started and maintains UDT in pain management along with fear. Fear of prosecution has been attributed to the proliferation of drug testing by doctors treating chronic pain. Goldberg and Rich write, “This singular focus strongly suggests purposes beyond ensuring quality patient care, such as fear of regulatory scrutiny and potential legal liability.”
The CDC has turned a blind eye to a number of important issues regarding UDT in pain management, including its constitutionality and the fact that it was likely driven by profits. Group think can negatively influence treatment guidelines and that is likely to blame for the CDC urine drug test recommendation. Giving a person with pain a “choice” to either submit to a drug test or not receive pain medication is really the option to either submit to a search or suffer, and that is coercion. It exemplifies patriarchal medicine at its worst and is the antithesis of patient-centered care.
James L. Madara, MD, CEO of the American Medical Association, was quoted as saying about the CDC guideline, "The guidelines and supporting discussion are devoid of a patient-centered view and any real acknowledgment or empathy of the problems chronic pain patients may face."
The CDC should not recommend UDT as part of the current guideline since there is no proof of efficacy, it may be unconstitutional, and was likely driven by profits and nurtured through fear. Furthermore, it is very expensive and may cause harm to the patient and patient-provider.
Mark Collen is an independent scholar and patient advocate. He serves on the editorial board of the Journal of Pain & Palliative Care Pharmacotherapy, and has peer reviewed manuscripts for journals including The Patient: Patient-Centered Outcomes Research, The American Journal of Pharmacy Benefits, and The Clinical Journal of Pain.
Mark is also the founder of PainExhibit.org, an online art exhibit from artists with chronic pain, which seeks to educate healthcare providers and the public about chronic pain through art, and to give a voice to the many who suffer in silence.
This column is an abbreviated version of the comment Mark has submitted to the CDC. The full version of Mark’s comment can be read here.
The information in this column should not be considered as professional medical advice, diagnosis or treatment. It is for informational purposes only and represents the author’s opinions alone. It does not inherently express or reflect the views, opinions and/or positions of Pain News Network.