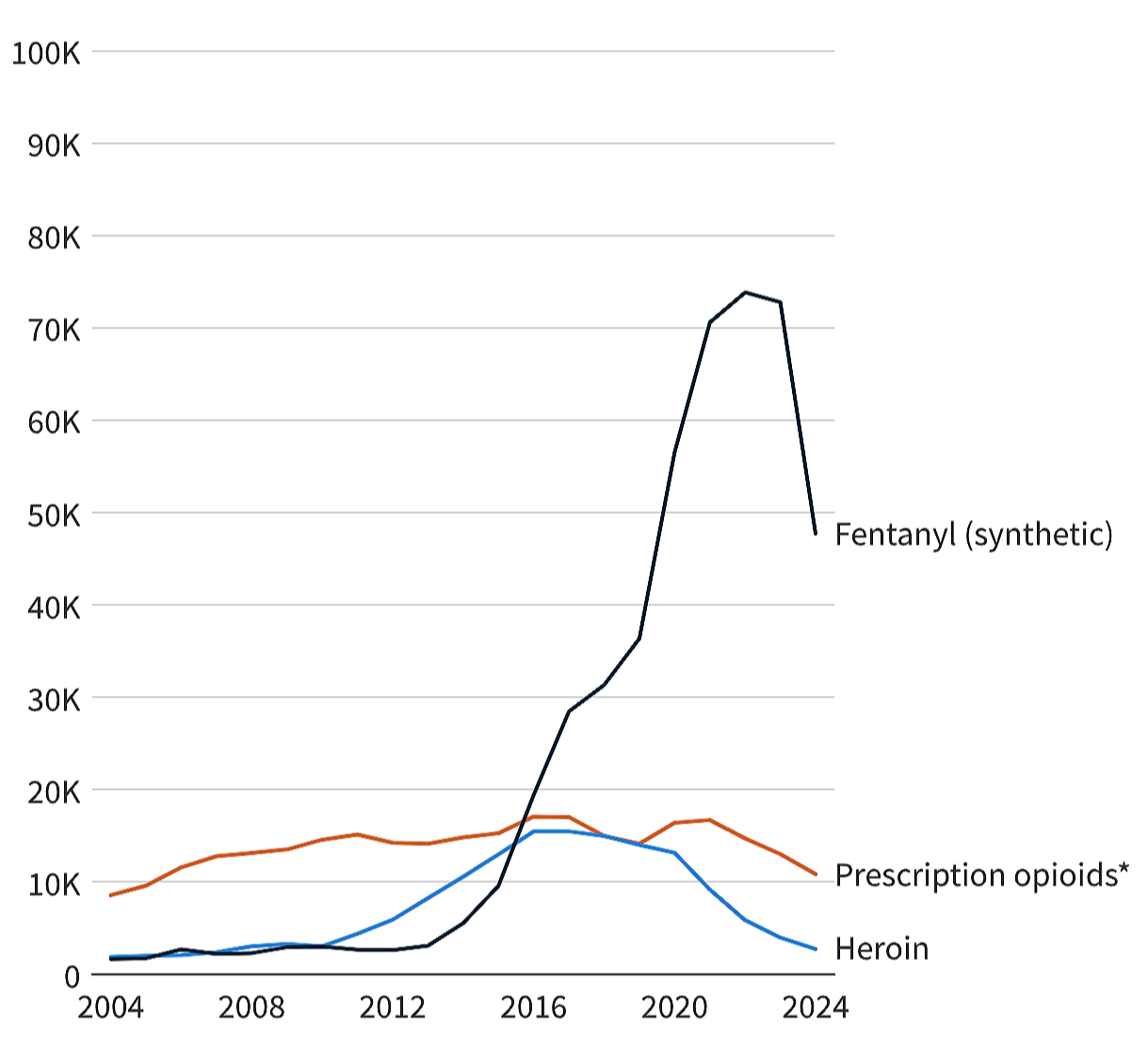
A Simple Chart Destroys a Myth About the Opioid Crisis
/By Neen Monty
There’s a simple reason the narrative around the “opioid crisis” falls apart when you actually look at the overdose data.
Because the data doesn’t show what they claim it shows.
And that false narrative has been retrofitted to cover the deception and lies wrought by bad actors in the U.S. healthcare system.
The chart below tracking overdose deaths by opioid type tells two stories:
Prescription opioid deaths rose, stabilized, and then fell.
Fentanyl arrived… and everything exploded.
Those are not the same curve. Those are not the same problem.
There are two epidemics. And one deliberately blurred narrative.
In the United States, prescription opioid deaths peaked around 2012, at about 5 deaths per 100,000 Americans.
Meanwhile, fentanyl-related deaths – almost non-existent before 2012 – began rising exponentially as illicit fentanyl entered the black market.
In 2023, they peaked at 22.2 deaths per 100,000 people. By then, overdoses linked to prescription opioids had fallen to 3.8 deaths per 100,000.
That means there are almost 6 times the number of overdose deaths caused by illicit fentanyl than prescription opioids.
Yet prescription opioids remain the target.
That “opioid crisis” curve?
That’s illicit fentanyl. Not pain patients. Not prescription opioids. Not doctors treating disease.
The Uncomfortable Truth
When overdose deaths skyrocketed, prescription opioid deaths were already flattening out and falling. That should have changed the narrative right there.
But the narrative stayed the same.
The explosion in deaths aligns with one thing: iIlicit fentanyl and its analogues flooded the U.S. drug supply with counterfeit pills of unknown, often lethal, potency.
That is why deaths surged, not because someone with intractable rheumatoid arthritis got oxycodone prescribed for their pain.
The numbers don’t lie. If the opioid crisis were really about prescriptions, the lines on the chart would look very different.
So why didn’t the narrative charge?
Because blaming illicit fentanyl means:
Confronting illicit drug supply chains
Admitting policy failure
Acknowledging complexity
Blaming prescriptions is easier. It creates easy villains to target:
Doctors
Patients
Pain medication
And it opens the door to:
Restrictions
Guidelines
“Education programs”
Entire industries built on fear
Who paid the price?
Pain patients. People with cancer, autoimmune disease, neurological damage, and severe structural pathology.
People who were stable. Functioning. Living.
Until they were cut off.
This was never about prescriptions. The chart makes one thing brutally clear: The U.S. overdose crisis is a fentanyl crisis.
Everything else is a false narrative. A fairy story. One designed to vilify people who are already among the most vulnerable people in our communities – the ill, the disabled, people living with severe pain.
It’s important to note that by the time Physicians for Responsible Opioid Prescribing (PROP) was created in 2012, prescription opioid deaths had already stabilised and were falling.
PROP was instrumental in creating the CDC’s infamous 2016 opioid prescribing guidelines. But there was no need to restrict opioid prescribing. There was no need for forced tapers that lead to suicides and overdose deaths. There was no need for dose ceilings that meant people’s pain was no longer treated adequately.
There was also no need for the fabrication of “evidence” that does not stand up to even a little scrutiny. Just read the studies that the CDC guidelines are based on. They do not show what you have been told they show.
But no one actually reads the studies. No one examines the data. No one questions. Doctors just blindly follow the guidelines. It’s hard to blame them when they face prison time if they don’t.
It has been ten years since the forced tapers started in the U.S. Six years since Australia blindly copied this failed policy. No one in Australia bothered to read the research. Australia just copied the U.S. guideline and allowed politicians to decide who gets pain relief and who does not.
Australia could see the policy going horribly wrong in the U.S. Yet it implemented those same policies and tortured Australian chronic pain patients the same way. The same is true for Canada and the UK as well.
This should never have happened.
Patients on three continents have been abandoned, left to suffer in agony day after day. Given psychological therapies for physical disease and injury. And no one says a word.
I have contacted many politicians, journalists, and senior public servants in Australia. No one will take this on. No one will right this wrong.
And the media continues to push a false narrative about an opioid crisis that does not exist. But it gets clicks.
It may be futile, but my life and those of many others depend on access to safe and effective opioid therapy.
And so, I will continue to fight.
Neen Monty is a patient advocate in Australia who lives with rheumatoid arthritis and Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), a progressive neurological disease that attacks the nerves.
Neen is dedicated to challenging misinformation and promoting access to safe, effective pain relief. For more information on chronic pain, the science, the politics and the lived experience, go to Pain Patient Advocacy Australia. You can also subscribe to Neen’s free newsletter on Substack: “Arthritic Chick on Chronic Pain.”