Is the Vagus Getting On Your Nerves?
/By Madora Pennington
After writing about a nerve stimulation implant approved by the FDA to treat rheumatoid arthritis by targeting the vagus nerve, I just had to try neuromodulation myself.
While my condition is a genetic disorder, not autoimmune, there is a lot going wrong with me. Could a vagus nerve stimulator help me?
Neuromodulation devices send electrical pulses to nerves to alter their activity. These signals can inhibit pain, seizures or tremors. They can also stimulate neural impulses when they are deficient or absent, inducing positive changes in the body.
Luckily, many manufacturers are making non-invasive vagus nerve simulators that are available without a prescription, so I did not have to get anything surgically implanted. The devices send energy through the skin at points on the neck or ear, where the vagus nerve isn’t deep inside the body.
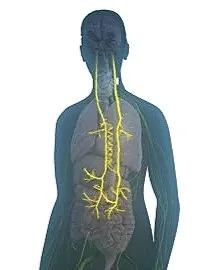
The vagus nerve is the longest nerve we have – originating inside the skull and traveling down through the neck, chest and abdomen. It interacts with many organs and the immune system, and carries important information back to the brain.
Signs that your vagus nerve may be underperforming include:
digestive problems
difficulty swallowing
dizziness or lightheadedness
fainting
slow recovery from illness or injury
anxiety
depression
poor emotional regulation
chronic fatigue or low energy
migraines
insomnia
I have Ehlers-Danlos Syndrome (EDS), an inherited collagen defect. EDS is considered a “high-burden” disease as it causes many debilitating symptoms that can be near impossible to alleviate.
The hallmark of EDS is unstable joints that bend backwards, but most of us who have it have many of the problems listed above. Could this be a sign of the vagus nerve not functioning correctly?
To find out for myself, I evaluated a number of devices. The good news is that most companies that sell vagus nerve stimulators offer a trial period during which the purchase can be returned for a refund, so you can see if it works for you.
I bought the Hoolest VeRelief Prime, a handheld device that sells for $199, because it does not run through a phone app. I spend more time than I want looking at my phone already, plus I did not want to be stuck paying for an app just to use an expensive item that I already purchased.
I also liked how compact the VeRelief Prime is. There are no wires or extraneous parts, no screens, just buttons.
Founder Nick Hool told me his interest in calming the nervous system started when he developed severe performance anxiety as a high school golfer.
“People told me just relax, breathe, do positive affirmations, go for a walk, all the stuff that is suggested for bad anxiety,” he said. “Those are good daily practices for general well-being, but they don't work on someone who's having a nervous system breakdown. You can't think your way out of a paralyzing fight or flight response."
I have to say, I agree. Various relaxation techniques can be soothing, as long as I don't need too much soothing.
In working on his PhD in biomedical engineering, Hool researched and tried vagus nerve stimulators on himself and learned they don’t usually work right away.
“You don't feel immediately better after one session,” said Hool, who recommends using a stimulator daily for at least six weeks to strengthen the vagus nerve and get results. Less frequent use will then maintain the improvements.
I, however, felt better immediately, and was in a calmer and brighter mood. The reduced stress and improved resilience continued to build as I used the device over weeks and then months.
Shortly after I got the VeRelief device, I took a cross-country trip. I'm not a great traveler, even though I want to be. Time changes, different beds, jet lag, lugging my luggage, and sensory overload all can be pretty aggravating. On top of that, sitting still on a plane for hours irritates my back. I usually come home miserable.
As I flew back on a late-night, crowded, delayed plane that hadn’t been cleaned, after a week of exhausting tourist activities, I was, much to my surprise, just fine. I discreetly took out my little device and held it against my neck when I began to feel discomfort.
Since then, I don't hesitate to bring the stimulator with me when I know I will encounter a troublesome environment, such as pain triggers, bright fluorescent lights, and difficult people. If other people ask, I tell them it's to prevent migraines.
The device is small and silent. No one seems to mind. I even use it during psychotherapy. It seems to help me process uncomfortable topics quickly.