Canada Seeks Feedback on Proposed Opioid and Cannabis Guidelines
/By Pat Anson, PNN ditor
Health officials and pain management experts in Canada are seeking public feedback on proposed changes to Canada’s 2017 opioid prescribing guideline. They’re doing it through an online survey, with responses accepted not only from Canadians, but from providers, patients, caregivers and policymakers in the United States and other countries.
“We are very happy to receive feedback from both Canadians and Americans,” says Jason Busse, DC, a clinical epidemiologist and professor of anesthesia at McMaster University. “We are still in the process of drafting the guidelines, as the feedback we receive will affect these documents.”
Busse is leading a panel at McMaster’s Michael G. DeGroote National Pain Centre that has developed 11 draft recommendations for a revised opioid guideline. Survey respondents are being asked whether they agree or disagree with the recommendations, which focus on whether opioids should be a first line treatment for people with chronic non-cancer pain.
Consistent with the current guideline, the panel believes there are several non-opioid treatments that are just as effective as opioids and have less risk of adverse events. It also strongly recommends that opioids not be given to people with a history of opioid overdose, substance abuse, or mental health problems. For pain sufferers without such a history, the panel recommends “discussing a trial” of opioids if nonopioid treatments haven’t lessened their pain.
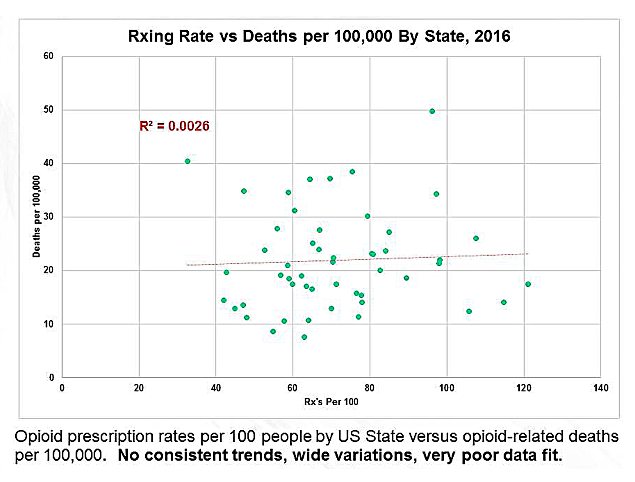
If opioids are prescribed, the panel suggests avoiding daily doses higher than 80 morphine milligram equivalents (MME), and strongly recommends against doses higher than 150 MME. The estimated risk of a fatal overdose at 80 MME is relatively low at 0.23% (2.3 overdoses for every 1,000 people) and at 150mg MME is 0.5% (5 in 1,000).
The 80/150 MME recommendations are a substantial change from Canada’s current guideline, which suggest that initial doses of opioids be less than 50 MME and that they not exceed 90 MME.
For people in pain who are stable on long-term opioid therapy, the panel recommends that clinicians “initiate a discussion” of tapering to a lower dose, potentially including discontinuation. If the patient refuses, the panel recommends that another “discussion” be repeated every 6 to 12 months. Forced or involuntary tapering is not recommended.
“The Guideline Panel has formulated their recommendations based on current evidence and the values and preferences of people living with chronic pain. We are seeking public feedback on the current wording of the recommendations and associated remarks. We will review all feedback received in order to further optimize the wording and clarity of the recommendations and remarks,” Busse said in an email to PNN.
To take the opioid survey, click here.
Medical Cannabis Survey
A second survey is also being conducted by the National Pain Centre to help formulate a Canadian guideline for treating chronic pain with medical cannabis. As with the first survey, this one is open to everyone, regardless of where they live.
There are currently four draft recommendations for cannabis. The panel recommends a “trial of cannabis” for chronic pain only if a patient has tried other therapies that haven’t worked. The panel says there is evidence of small improvements in pain, physical function and sleep when cannabis was used.
Smoking cannabis is not recommended. The panel suggests that patients start with low doses of cannabis taken orally in oils and soft gels or inhaled through a vaporizer. Doses can be increased, depending how a patient responds.
Another recommendation is that cannabis be used by patients as a substitute for opioids or to help taper them to lower doses. The panel says “there is little to no difference” in effectiveness between cannabis and opioids, but that cannabis has far less risk of adverse events.
To take the cannabis survey, click here.
Both surveys will take about 20 minutes to complete and will remain open until 12pm ET, February 29, 2024.