By Neen Monty
The current myth in opioid science is that buprenorphine is inherently safer than oxycodone.
The truth is far more ordinary: it depends on the individual. Some people do better on buprenorphine. Others do better on oxycodone. There is no universal “safer” opioid for every patient.
I have trialed buprenorphine before. It was profoundly sedating. I could not function on it.
Despite this, I was required to trial it again.
Here’s what happened.
Week One: Hope
The first few days were incredible.
Less pain.
More functional hours.
And I was sleeping until 5am!
I was thrilled to be wrong about buprenorphine. I ignored the severe nausea and the nagging headache because I could go back to the archery range. I started planning a fitness routine. I allowed myself to feel hopeful.
Week Two: Wearing Down
The nausea intensified. Even with ondansetron (Zofran), it was relentless.
The headache persisted. Not dark-room migraine territory, but just constant enough to make thinking difficult.
Panadol. Nurofen. Naproxen. Nothing touched it.
I also realised my mood had shifted. I felt low. Irritable. Short-tempered. Sad. Defeated.
I controlled it. I always do. I don’t take out my moods on others. My children didn’t see it. They saw normal. They did not see the enormous effort it took for me to stay calm and reasonable.
The crying happened in the one private space I have: the bathroom. The sadness stayed hidden.
Week Three: Something Was Wrong
By week three, I was done.
My pain doctor told me to persevere. I did, as long as I could. At the end of week three, I ripped the buprenorphine patch off and did not apply the next one.
Enough was enough.
Within four days the nausea was gone.
The headache was gone.
I could think again. I could function again.
You never realise how severe something is until it stops.
Why did I tolerate it for so long? Because if I am labelled “non-compliant,” if I am judged not to be trying hard enough, I risk being forcibly tapered off all opioid medication.
And that would be catastrophic for me.
So, I tried.
Positive attitude! Yes, Doctor. I will trial it. With a smile on my face.
But I cannot continue with buprenorphine.
Next: Palexia
Now the rotation moves to another opioid: Palexia (tapentadol).
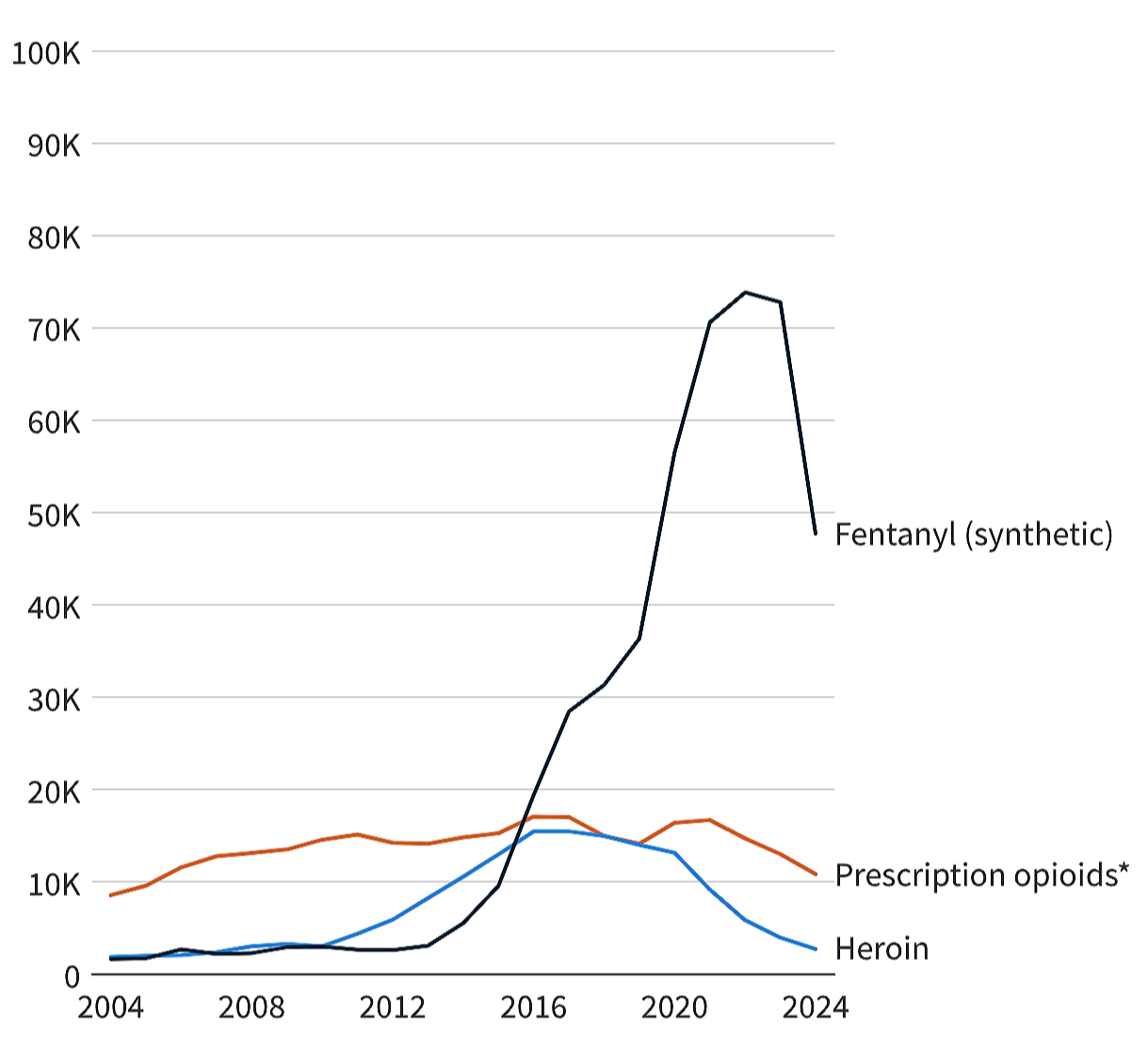
Doctors have been convinced that these newer opioids are “safer” than oxycodone. There is no high-quality evidence showing they are safer for stable, long-term chronic pain patients. But the safety narrative has been repeated so often, it is treated as fact.
Palexia is now the most commonly prescribed opioid in Australia. It has overtaken oxycodone. This did not happen because Palexia offers improved pain control. Or because it has fewer side effects. Or because it’s safer.
It happened because doctors were encouraged — in some cases pressured — to rotate patients off their “old school” opioids, to “atypical opioids” like Palexia, buprenorphine, and even tramadol.
A patient’s preference is no longer central. Being stable on a dose is no longer enough.
Opioid rotation is being enforced on me for two reasons.
First, because policies and guidelines position buprenorphine and tapentadol as the “safest” opioids. This is not evidence based; it is opinion.
Second, because I am not getting enough pain relief from my current regimen. My 20mg of oxycodone stops working after 6-8 hours, when it should last 12 hours.
The obvious solution is to prescribe it every 8 hours, instead of every 12 hours. That’s what happened in the past and was the standard of care. Because it is well recognised that the 12-hour formula rarely lasts 12 hours.
But that would put me over the arbitrary daily dose ceiling of 100 MME. Which is also not evidence based.
What is evidence based is that I was happier, healthier, fitter and more functional on 120 MME, rather than 100 MME.
But my well-being, pain control and function are not the important issues here.
Instead of the simple and obvious solution, the only solution is opioid rotation. The theory being that I have built up tolerance to oxycodone, but I have not built up tolerance to buprenorphine or tapentadol. So those medications will supposedly work better for me.
Except that’s not how it works in the real world.
Pain relief is not the goal here. It’s just not that important. It’s a money-making policy masquerading as “safety.”
Follow the money if you want to understand rapid prescribing shifts. Pharmaceutical policy rarely moves without financial incentive.
But here is the uncomfortable truth: My GP is not corrupt. She is not malicious. She is a very good GP. She is following guidelines.
She does not have time to audit every citation behind every recommendation on every guideline for every condition she treats. She has to trust her medical college, her training, and the documents placed in front of her.
That trust is not deserved. At least when it comes to treating chronic pain.
The current Australian guidelines are built on expert opinion, not randomised controlled trials. Opinion, layered over selective and cherry-picked evidence. Shaped heavily by academic pharmacology and population-level policy concerns. Not patient outcomes, preference, function or stability.
And patients like me pay the price.
A Four-Week Pause
I told my GP I cannot continue buprenorphine.
She immediately began discussing Palexia.
I asked for four weeks. Just four weeks to stabilise. I am only just beginning to feel like myself again. I am only just becoming functional again. I just lost four weeks of my life to the last rotation attempt. I don’t feel ready to tackle the next medication yet.
I need to take a breath. I need to get back to “normal.” Albeit my normal.
This rotation destabilised me. Made me far worse, not better.
Also, it would make medical sense to start the next medication from a steady baseline, not during biochemical and emotional turbulence.
She agreed. Very readily. I didn’t have to argue my case. I very much appreciate that. She prescribed my usual regimen of oxycodone extended release and immediate release.
So, for four weeks, I remain on oxycodone. For four weeks, I will stabilise. And feel like myself again.
After that, the forced rotation begins again. I have trialed Palexia before, a few years ago. It did not go well. But still, I am being forced to trial it again.
Round and round.
None of this is my choice.
None of this is evidence based.
None of this is for my benefit.
The Systemic Problem
This is not just about me.
Patient choice has been steadily displaced by policy-driven prescribing. Stability is no longer considered sufficient. Long-term patients who are functioning are being told their medication is “unsafe.” Not because of their individual outcomes, but because of population-level risk narratives built on low-quality evidence and relative risk framing.
Meanwhile, newer opioids are positioned as “safer” without robust comparative long-term data in stable chronic pain populations.
When prescribing patterns shift this dramatically and this quickly, it is reasonable to ask whether evidence alone is driving the change.
Medicine should be individualised. Instead, it is being standardised around risk optics, regulatory anxiety, and market forces.
And patients bear the instability.
Yes, some patients can safely be rotated, with no adverse events. But not all patients.
A large percentage are objectively and substantially worse off. But this does not matter. Patient satisfaction, patient function, pain relief are all secondary concerns. Often not considered at all. Or, if acknowledged, are dismissed as the patient being difficult or non-compliant.
Patients no longer have any agency, any choice, any control, or even a voice.
Neen Monty is a patient advocate in Australia who lives with rheumatoid arthritis and Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), a progressive neurological disease that attacks the nerves.
Neen is dedicated to challenging misinformation and promoting access to safe, effective pain relief. For more information on chronic pain, the science, the politics and the lived experience, got to Pain Patient Advocacy Australia. You can also subscribe to Neen’s free newsletter on Substack, “Arthritic Chick on Chronic Pain.”